This is a very interesting observation. I might have an explanation but one that doesn’t really provide a solution beyond what
@Freshflora mentioned regarding ditching the pH for controlling CO2.
In my line of work I have to accurately measure pH of various water sources. High purity RO/DI, steam condensate, waste streams, cooling towers, boilers etc. all with varying levels of ionic strength. For the ultra pure water sources, I need to have a dedicated pH probe calibrated with low ionic strength buffers.
Ionic strength effects the pH, google Henderson–Hasselbalch equation if you want a brain cramp for a while. It’s been over 20 years since I had to actually use that stuff (thanks advanced analytical chemistry) so it would take me a while to explain it properly.
Most readily available pH buffers are high ionic strength. Measure the TDS of your buffers and compare it to your tank water. You should see a large difference. That large difference can lead to you pH probe calibration being way off from the true pH.
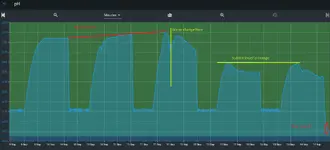
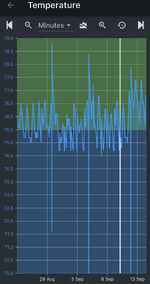
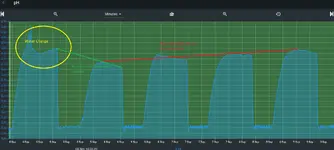
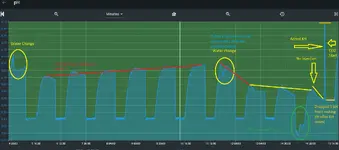
That in and of itself isn’t a problem. As long as the curve is wrong at the top and bottom the pH controller should be close enough. The problem arises as things change over the course of a week. Rising or falling TDS charges the ionic strength of the tank and therefore changing the pH the controller is calculating. For non RO/DI tanks it probably isn’t noticeable but for tanks with minimal reconstitution and lean dosing it could be a large enough change to throw off the pH and thus your set point. I did see you use RO/DI but didn’t see your dosing/reconstitution amounts.
Seneye link this explains it well without the chemistry mumbo-jumbo I threw in above. It has some marketing stuff in it but it’s a decent explanation.
I could be way off on all of this explanation but I wanted to mention it. I did notice with my tank since I stopped using my pH controller and just injecting at a constant rate and only monitor the pH with the controller plant health and fish behavior has improved significantly.