Chapter 2: Growing plants with lean fertilisation in the water column
Introduction
See Chapter 1 for an introduction and more general information about this experiment.
More detailed information and further experiments can be found [if interested] on my website: golias.net/akvaristika/.
Plants
In this experiment I decided to use the following emersion plants (i.e. plants grown in a greenhouse):
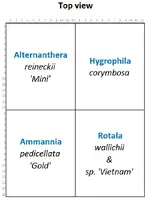
Diagram of plant placement in individual aquariums ↓
Picture of the fifth aquarium (few days old) ↓

Light
Lighting interval: 8h/day
Light intensity (PAR) in individual aquariums: 100-230 µM/m2·s (bottom to top)
Substrate
While in the first set (aquaria #1 to #4) a soil substrate was used, in the second set (aquaria #5 to #8) no substrate was used.
1st set → nutrient-rich substrate


Note: The recipes below were prepared using pure (demineralized/deionized) reverse osmosis water.

planting: 2024-12-18


week #1: 2024-12-21


week #2: 2024-12-28


week #3: 2025-01-04




week #5: 2025-01-18


To be continued ...
Introduction
See Chapter 1 for an introduction and more general information about this experiment.
More detailed information and further experiments can be found [if interested] on my website: golias.net/akvaristika/.
Plants
In this experiment I decided to use the following emersion plants (i.e. plants grown in a greenhouse):
- Alternanthera reineckii 'Mini'
- Ammannia pedicellata 'Gold' (formerly known as Nesaea sp. Gold)
- Hygrophila corymbosa
- Rotala wallichii & Rotala sp. 'Vietnam'
Diagram of plant placement in individual aquariums ↓

Picture of the fifth aquarium (few days old) ↓

Light
Lighting interval: 8h/day
Light intensity (PAR) in individual aquariums: 100-230 µM/m2·s (bottom to top)
Substrate
While in the first set (aquaria #1 to #4) a soil substrate was used, in the second set (aquaria #5 to #8) no substrate was used.
1st set → nutrient-rich substrate

- I used the ADA Aquasoil Amazonia substrate (in powder version), which I soaked in tap water for two weeks before starting the experiment.

- In aquariums with no substrate, I used M16 stainless steel hex nuts inserted into small size (2") hydroponic net pots to anchor (secure) the plants.
Note: The recipes below were prepared using pure (demineralized/deionized) reverse osmosis water.

- Water flow ensured by a surface skimmer (Jingye JY-350)
- no filtration used
- Water changes done once a week (with 50% of the water changed) with macro-nutrients replenishment
- Micro-nutrientsadded every other day
- Weekly amount of microelements (divided into three doses):
- Fe/DTPA = 0.04 ppm (+ 0.02 ppm Fe/gluconate)
- Mn/DTPA = 0.02 ppm
- B = 0.01 ppm
- Zn/DTPA = 0.006 ppm
- Cu/DTPA = 0.002 ppm
- Co/DTPA = 0.00002 ppm
- Mo = not added
- Weekly amount of microelements (divided into three doses):
- Carbon dioxide:
- Extra CO₂added to aquaria #1, #3, #5 and #7 using a simple glass diffuser, the function and parameters of which are described in more detail in a separate article
- CO₂ concentration in these aquaria: ~11 ppm
- No extra CO₂added to aquaria #2, #4, #6 and #8
- CO₂ concentration in these aquaria: ~4 ppm
- CO₂ concentration measured by Carbon Dioxide Chemical Test Kit (Hanna HI3818), which should give similar results to a professional CO₂ meter (OxyGuard) → see 2hraquarist.com.

- Hanna CO2 chemical test kit HI3818
- Extra CO₂added to aquaria #1, #3, #5 and #7 using a simple glass diffuser, the function and parameters of which are described in more detail in a separate article
planting: 2024-12-18


week #1: 2024-12-21


week #2: 2024-12-28


week #3: 2025-01-04


- Some fully grown or dying plants have been removed (so you may not see them in the following pictures).


week #5: 2025-01-18


To be continued ...
Last edited:













































